
Reductively cleavable linkers based on azo-dyes
Many biological and chemical applications require stable molecular bonds that can be cleaved gently, rapidly, and selectively under specific conditions. To this end, cleavable linkers are incorporated into the molecules, e.g. as a phosphoramidite building block in oligonucleotide synthesis. Common types of cleavable linkers include the PC linker (photocleavable linker) and disulfide linkers. Cleavage of the former requires additional irradiation equipment, whereas for the later, the linker often does not withstand the synthesis conditions.
Azo-dye-based cleavable linkers offer an alternative. Diazenyl-groups can be easily cleaved reductively under mild conditions, e.g. by treatment with dithionite (e.g., sodium dithionite, Na₂S₂O₄).
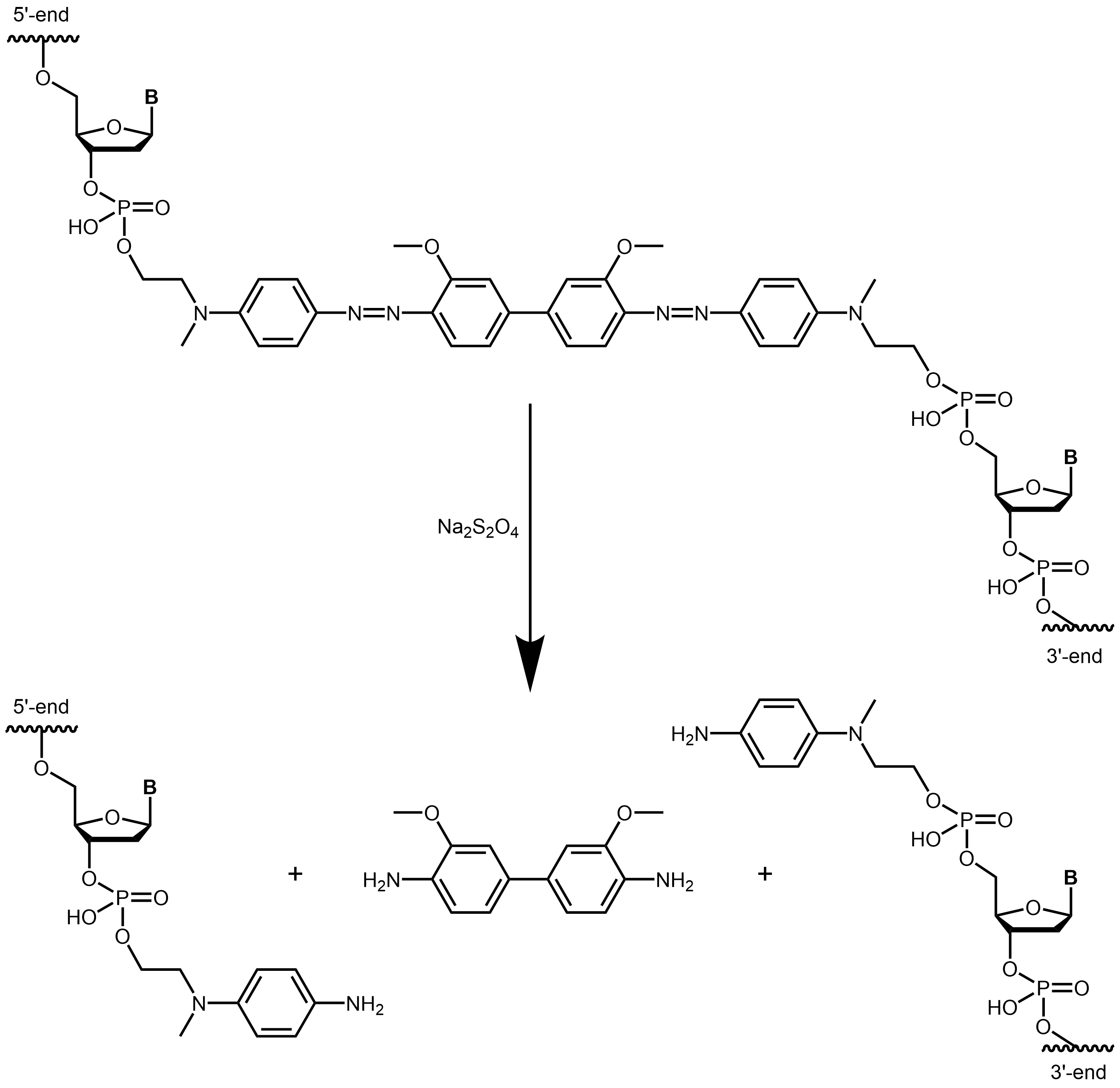
With the BMN-Q521 quencher as an internal modification, a reductively cleavable quencher is incorporated directly into the backbone of the oligonucleotide. As soon as the linker is cleaved e.g. with dithionite, the oligonucleotide is split and the quenching property of BMN-Q521 is lost. Any dyes coupled to the oligo can now indicate successful cleavage.


